On October 11th, the journal Cell Metabolism published a new article entitled "Protein Kinase C Epsilon Deletion in Adipose Tissue, but Not in Liver, Improves Glucose Tolerance", revealing a key enzyme, protein kinase. Metabolic details of C ε (protein kinase C epsilon, PKCε) in diabetes. The study was completed by Carsten Schmitz-Peiffer, associate professor at the Garvan Medical Institute in Australia, and his team.

An amazing discovery
PKCε is a cytoplasmic enzyme that regulates glucose metabolism. Past studies have suggested that PKCε is active in liver tissue and inhibits insulin by phosphorylating insulin receptors.
However, recent research has revealed that the main source of this key molecule contributing to diabetes is not the liver or the pancreas.
Often, scientists use a high-fat diet (HFD) to induce type 2 diabetes in mice, causing most mice to become "glucose intolerance" -- unable to control blood sugar after eating. In particular, the liver becomes "insulin resistant" - no longer responding positively to insulin.
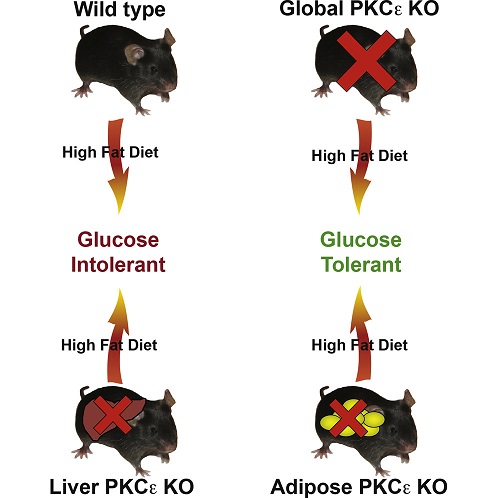
10 years ago, Carsten Schmitz-Peiffer found that PKCε is important for the development of diabetes. They confirmed that mutant mice that did not express PKCε at all did not develop symptoms similar to diabetes, even though they were in an environment where other normal mice were susceptible to diabetes.
"We found that mice that did not express PKCε at all did not develop glucose intolerant even with high-fat foods," Carsten Schmitz-Peiffer said.
To their surprise, when the PKCε expressed in the liver was removed, these mutant mice did not develop diabetes as in the past theory. In the latest study, these mutant mice had high blood sugar intolerance after fat feeding.
A new answer: fat
If it is not the liver, where does PKCε contribute to glucose intolerance?
Recent studies have found that when PKCε produced in adipose tissue is completely eliminated, glucose intolerance does not occur in mice. This means that the workplace where PKCε contributes to diabetes is not the liver but the adipose tissue.
PKCε is expressed in multiple tissues, but the latest research has given a new function to adipose tissue and further explains the link between obesity and high risk of diabetes.
Variety
Recent studies have found that fat cells show differences in shape and size when and in the absence of PKCε.
“Under the microscope, fat cells can be very different,” says Carsten Schmitz-Peiffer. “When adipose tissue does not express PKCε, most fat-fed mouse fat cells are small and healthy. Conversely, high expression of PKCε Fat-fed mice (with glucose intolerance), their fat cells are mostly unhealthy, swollen, difficult to obtain oxygen and prone to inflammation."
Carsten Schmitz-Peiffer believes that this change may have a wide and complex impact on diabetes. “We know that adipose tissue not only stores fat,” he explains. “It is a very active organ that releases many signals/molecules to other tissues in the body. PKCε affects the health of fat cells, which means it will further change fat. The signals transmitted by the cells, the released molecules, which play a role in organ tissues such as the liver, which interfere with glucose metabolism."
Inhibition of PKCε
This latest study provides new clues for drug development by targeting PKCε for the treatment of diabetes. Professor Schmitz-Peiffer is working with Professor Ray Norton and Professor Jonathan Baell of the Monash Institute of Medicine to develop an oral peptide based on a drug discovery platform for inhibiting the activity of PKCε.
References:
Protein Kinase C Epsilon Deletion in Adipose Tissue, but Not in Liver, Improves Glucose Tolerance
Fat tissue may play a crucial role in the progression of diabetes, challenging long established notions
Souce: NovoPro 2018-10-17
